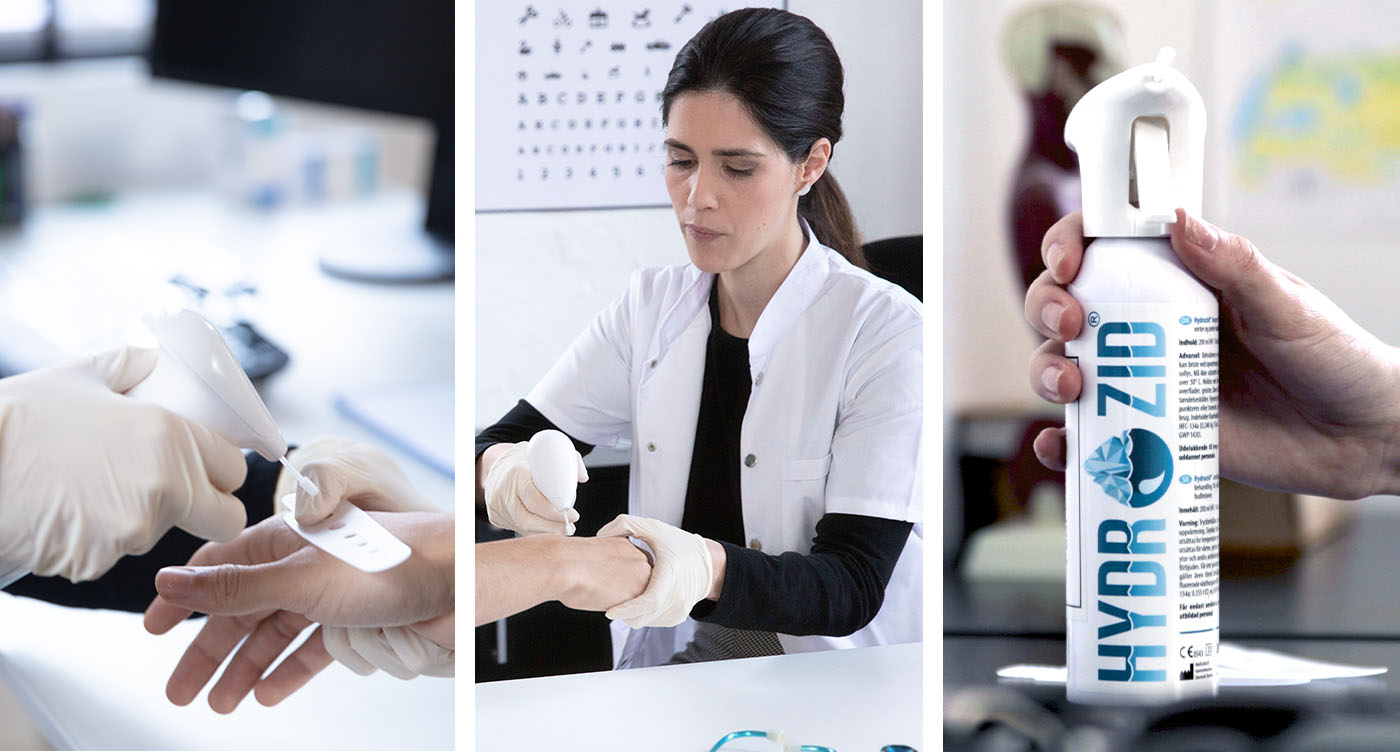
Hydrozid®, a leading cryosurgery device in Europe, is now FDA cleared and available to qualified providers in the U.S. Designed for primary care physicians, pediatricians, dermatologists, podiatrists, and other medical specialties, Hydrozid is a portable and convenient alternative to liquid nitrogen, with proven effectiveness in treating warts, seborrheic keratosis, actinic keratosis, and other pre-malignant skin conditions.
The device was developed in Denmark by BIBAWO Medical, a specialty pharma company. Originally sold to Danish dermatologists, the device is now sold to dermatologists, pediatricians, primary care physicians, gynecologists, urologists, and podiatrists, in 10 European countries. “Since 2015, we’ve experienced tremendous growth, as providers have experienced how easy and convenient Hydrozid is to use, and most importantly, how effective it is in treating warts and other skin conditions,” said Christian Kjerulf-Jensen, CEO of Medilink, BIBAWO Medical’s parent company. “We’re thrilled to have FDA clearance and be coming to the U.S., to avail providers of this unique device.”
The Hydrozid® system includes a 200mL light weight cannister, which keeps the pressure (and thus the temperature) consistent from the first use to the last. The Scandinavian design of the product, emphasizing functionality and ergonomics, makes it easy and comfortable to use. Hydrozid®’s unique spray head is patented in Denmark with a patent pending in the U.S. Templates provided with the system enable providers to select a hole matching the size of an individual lesion, thus protecting surrounding skin.
Since 2015, we’ve experienced tremendous growth, as providers have experienced how easy and convenient Hydrozid is to use, and most importantly, how effective it is in treating warts and other skin conditions
Hydrozid® utilizes norflurane, a medically approved gas that has no odor and is nonflammable. This propellant has been used for decades in asthma inhalers and has proven to be safe for patients. Used Hydrozid cans do not require special disposal.
BIBAWO Medical
BIBAWO Medical is a Danish specialty pharmaceutical company which produces, markets, and distributes medical devices and equipment. Headquartered in Bikerød, Denmark, the company also has offices in Portugal and India.